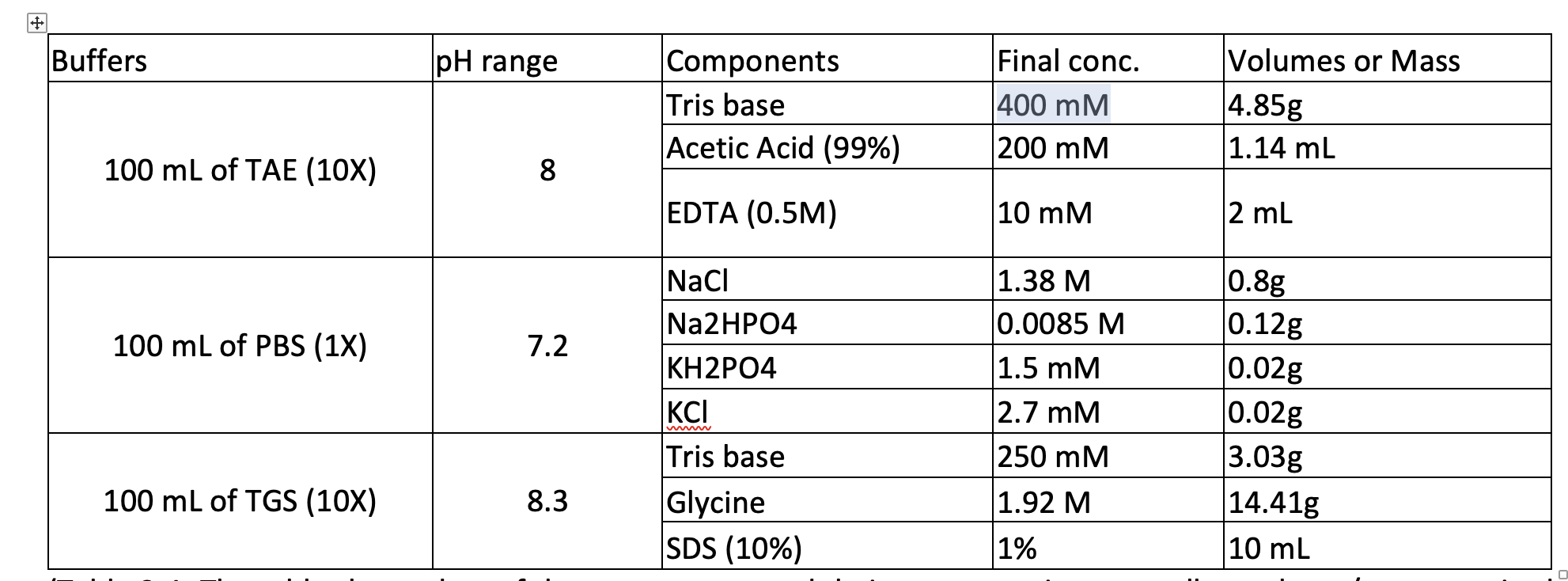
Measurement of pHT values of Tris buffers in artificial seawater at varying mole ratios of Tris:Tris·HCl | Semantic Scholar

SOLVED: Solution #1: 500. mL of 150 mM Tris, pH 8.50 You have the following available Tris base (FW = 121.1 g/mol) pKa 8.10) 1.0 M HCI Solution #2: 750. mL of

Tris(hidroximetil)-aminometano TRIS base is useful in the pH range of 7.0-9.0. Has a pKa of 8.1 at 25°C. | Sigma-Aldrich

SOLVED: Tris' is a buffer often used in biochemistry. We can simplify the equilibrium reaction as: Tris-HCl + H2O -> Cl- + H3O+ + Tris with pKa = 8.072 and the molar

WO2014165660A1 - Pharmaceutical formulations for subcutaneous administration of furosemide - Google Patents






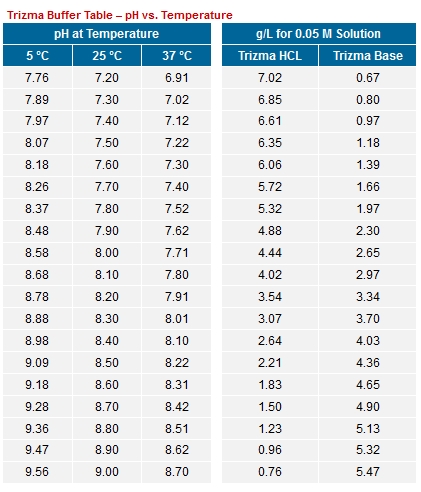

![BT156] 2M Tris-HCl, pH 9.5 | Biosolution BT156] 2M Tris-HCl, pH 9.5 | Biosolution](http://biosolution.cafe24.com/wp-content/uploads/2015/07/BT066-Tris-HCl.jpg)