![Brønsted base-catalyzed 1,2-addition/[1,2]-phospha-Brook rearrangement sequence providing functionalized phosphonates - Organic & Biomolecular Chemistry (RSC Publishing) Brønsted base-catalyzed 1,2-addition/[1,2]-phospha-Brook rearrangement sequence providing functionalized phosphonates - Organic & Biomolecular Chemistry (RSC Publishing)](https://pubs.rsc.org/en/Content/Image/GA/D2OB00256F)
Brønsted base-catalyzed 1,2-addition/[1,2]-phospha-Brook rearrangement sequence providing functionalized phosphonates - Organic & Biomolecular Chemistry (RSC Publishing)
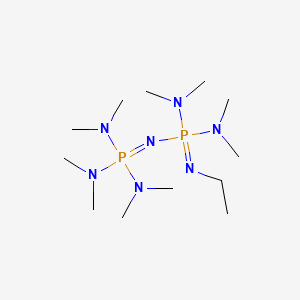
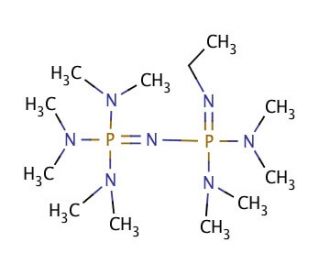
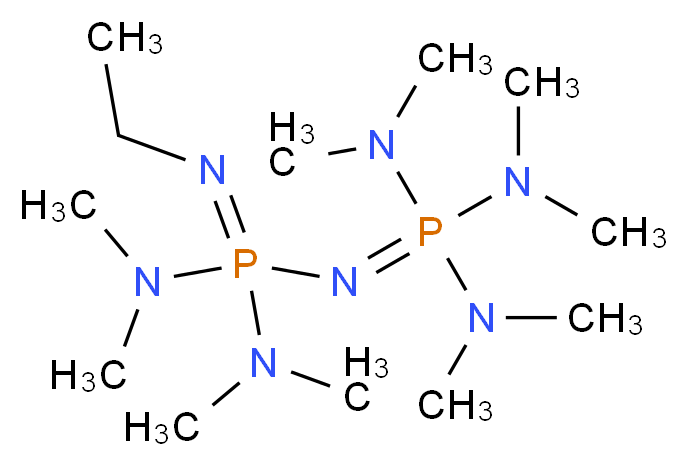
Phosphazene base-catalyzed condensation of trimethylsilylacetate with carbonyl compounds - Chemical Communications (RSC Publishing) DOI:10.1039/B606056K

P2Et Phosphazene: A Mild, Functional Group Tolerant Base for Soluble, Room Temperature Pd-Catalyzed C–N, C–O, and C–C Cross-Coupling Reactions | Organic Letters
![A Successful Application of Phosphazene Base P2‐tBu to [11C]ABP688 Radiosynthesis in Fully Automated Synthesis Module - Lee - 2020 - Bulletin of the Korean Chemical Society - Wiley Online Library A Successful Application of Phosphazene Base P2‐tBu to [11C]ABP688 Radiosynthesis in Fully Automated Synthesis Module - Lee - 2020 - Bulletin of the Korean Chemical Society - Wiley Online Library](https://onlinelibrary.wiley.com/cms/asset/8d307dde-94b3-4266-9dd3-2fbd42ebce38/bkcs12070-fig-0002-m.jpg)
A Successful Application of Phosphazene Base P2‐tBu to [11C]ABP688 Radiosynthesis in Fully Automated Synthesis Module - Lee - 2020 - Bulletin of the Korean Chemical Society - Wiley Online Library
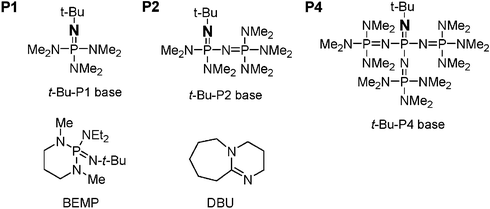
The phosphazene bases P 2-Et and P 4-tBu A more successful approach has... | Download Scientific Diagram

Organocatalytic Stereoselective Ring-Opening Polymerization of Lactide with Dimeric Phosphazene Bases | Journal of the American Chemical Society

P2Et Phosphazene: A Mild, Functional Group Tolerant Base for Soluble, Room Temperature Pd-Catalyzed C–N, C–O, and C–C Cross-Coupling Reactions | Organic Letters

P2Et Phosphazene: A Mild, Functional Group Tolerant Base for Soluble, Room Temperature Pd-Catalyzed C-N, C-O, and C-C Cross-Coupling Reactions. | Semantic Scholar