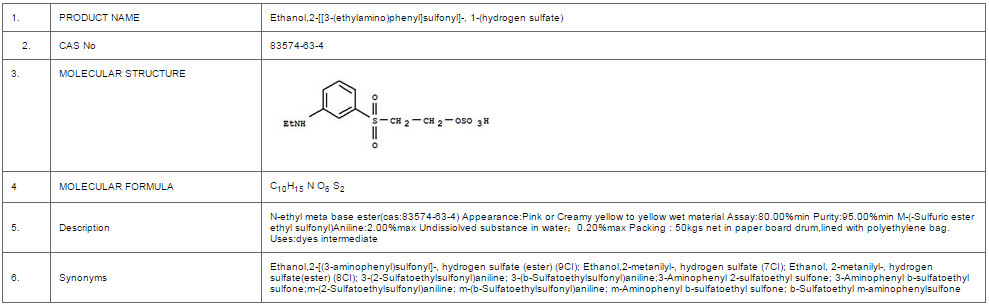
Photo-on-Demand Base-Catalyzed Phosgenation Reactions with Chloroform: Synthesis of Arylcarbonate and Halocarbonate Esters | The Journal of Organic Chemistry

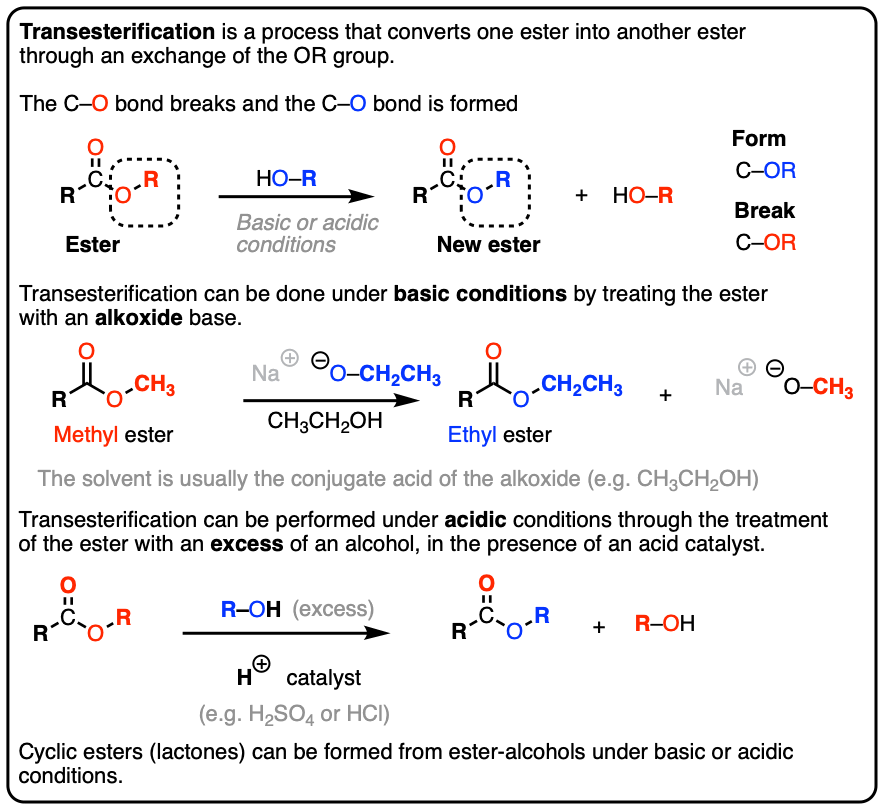
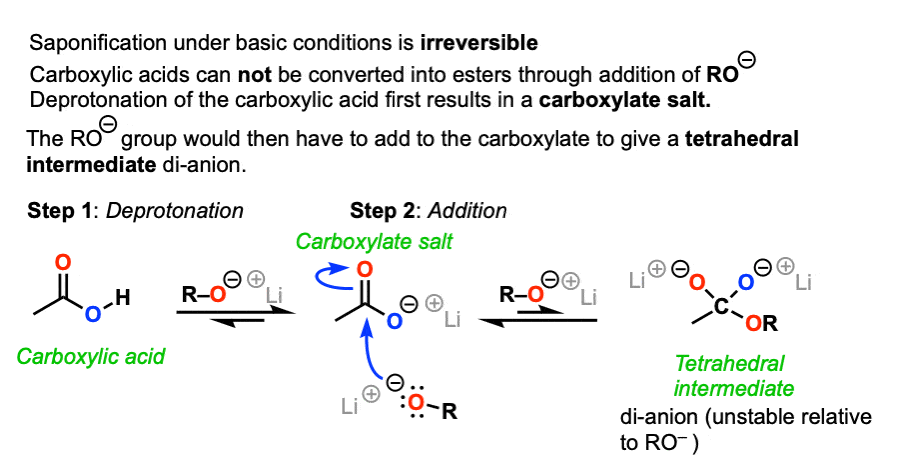
How can you convert a carboxylic acid into an ester? a. Heat with an alcohol and catalytic acid. b. Deprotonate with a base and react with an alcohol. c. Deprotonate with a
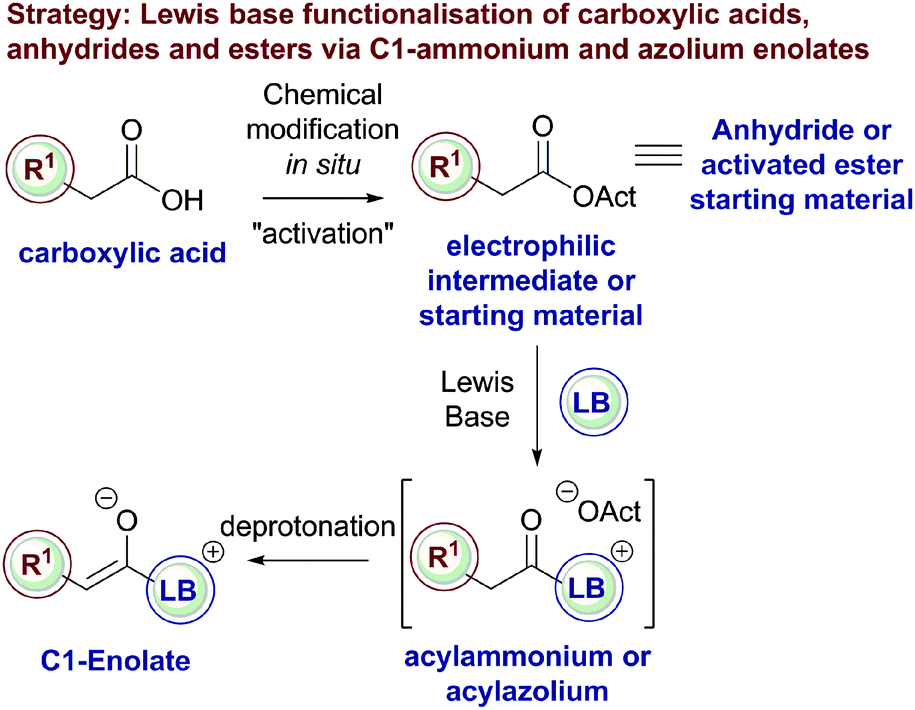
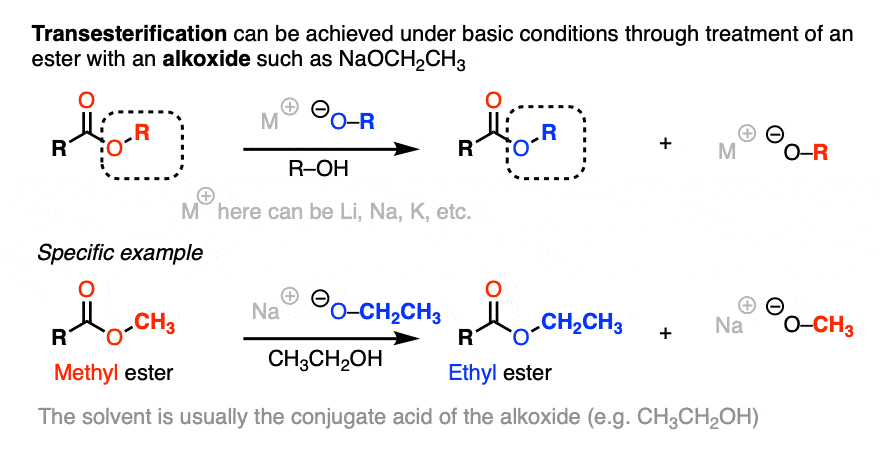


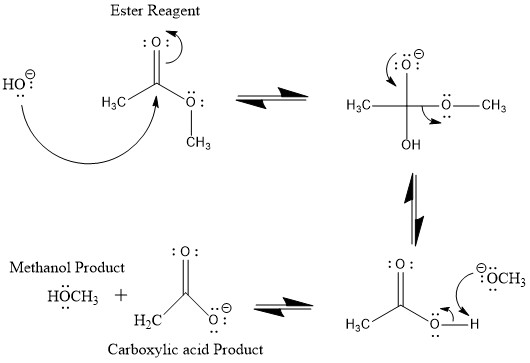

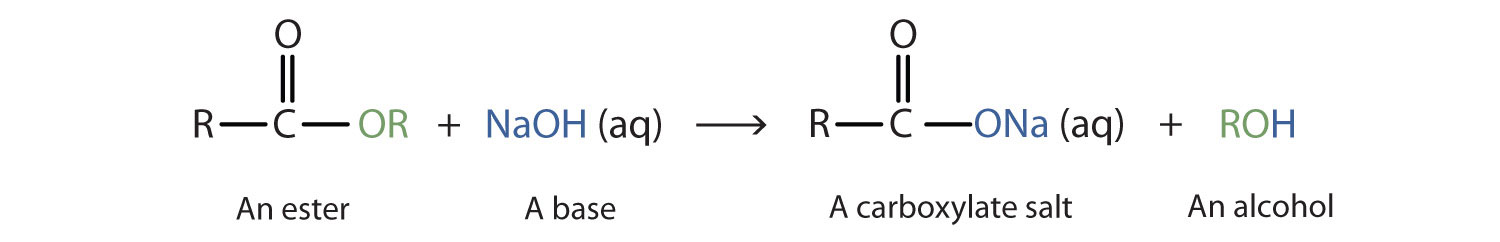
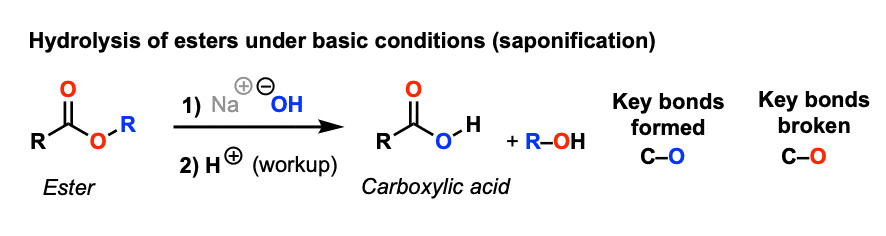
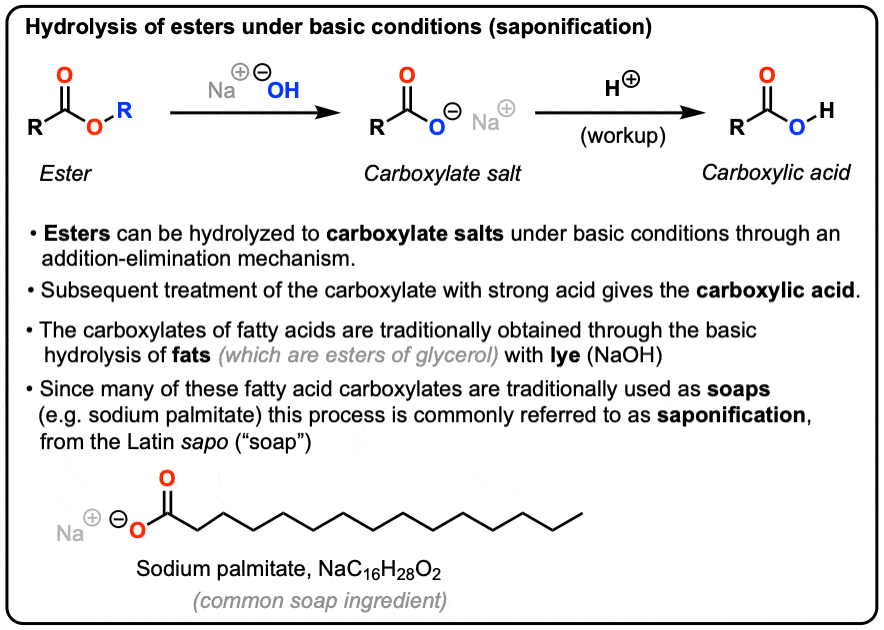

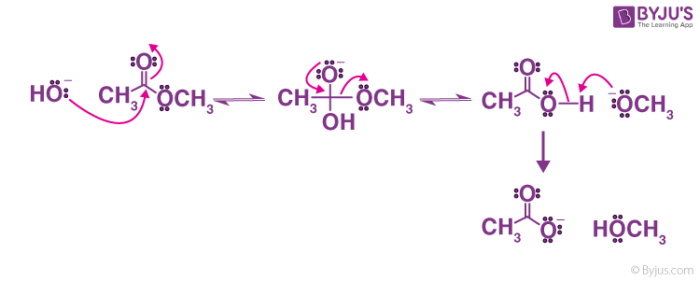
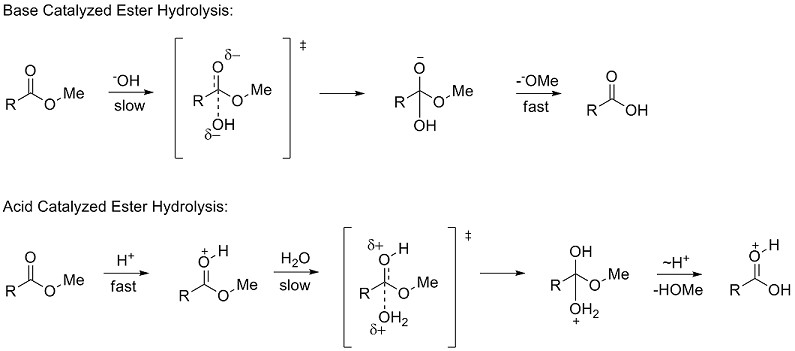
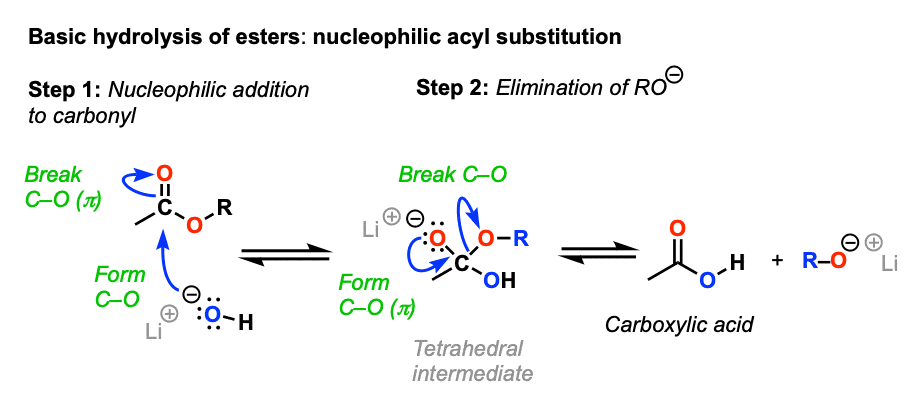
![Hydrolysis to carboxylic acid [basic] - ChemistryScore Hydrolysis to carboxylic acid [basic] - ChemistryScore](https://chemistryscore.com/wp-content/uploads/2019/11/Hydrolysis-to-carboxylic-acids-basic1-768x307.png)